Case studies
Explore our work
We’d love to talk you through the cool things we’ve done over the last 20 years! Here is a sample of a few ways we’ve applied UX and human factors research and design methods.
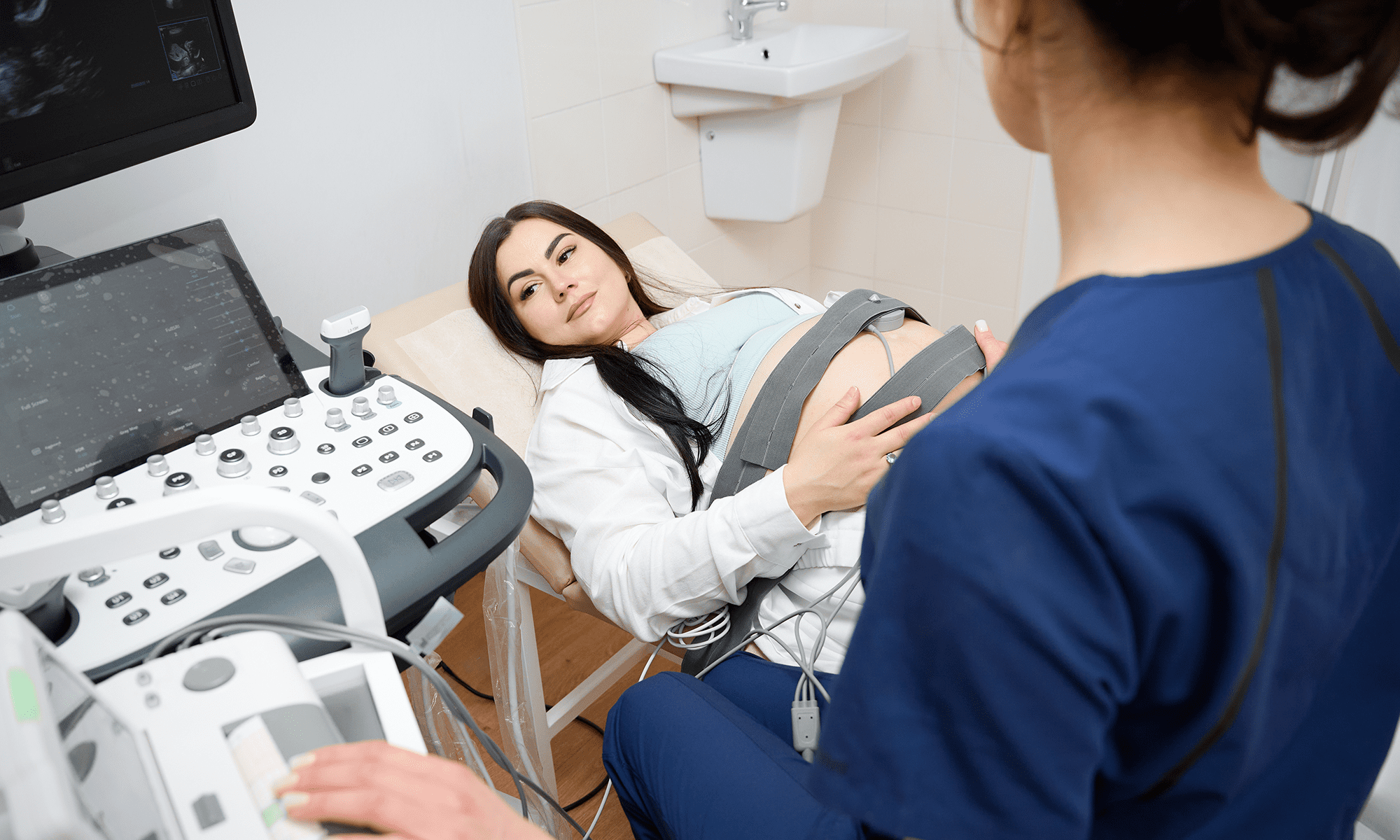
Our client set out to design an AI-enabled SaMD to assess pregnancy risk and predict adverse outcomes using patient data from primary and secondary care settings.
Build trust in AI-enabled SaMD with clinician-driven design

A multinational tech company needed to address low conversion rates on its app distribution platform in select Asian and South American markets.
Improving in-app payments across global markets

A global technology company needed to improve how they describe and categorize search tools (browsers, search engines, and virtual assistants).
Uncovering cultural differences in digital search behavior

Verizon engaged us to conduct one of the first large-scale research studies focused on cell phone usability for people who are blind or low vision.
Pioneering accessible mobile design for blind and low vision users

A global telecom company wanted to identify real, high-potential AR opportunities and shape a strategy that could help them lead in both mobile and wearable platforms.
Build a roadmap for growth in the augmented reality market

A medical technology company needed to confirm that a neonatal breathing circuit was ready for use in the US market and in its intended clinical environment.
Validating safe and effective use of a neonatal respiratory machine

A pharmaceutical company planned to bring a new drug delivery system to market. The system included an injector, instructions for use (IFU), training, and packaging.
End-to-end research program for new drug delivery system

A consumer products manufacturer was preparing to enhance an existing small home appliance with a new feature.
Aligning product design with consumer expectations for a new appliance feature

A fitness company was developing new metrics for treadmill workouts and wanted to understand how users interpret and value these features.
Drive feature adoption through user insights on treadmill workout metrics

A global pharmaceutical company set out to uncover how its US and Asia teams should best align for maximum productivity and output.
Diagnosing and improving global team dynamics

A diagnostics manufacturer wanted to understand how laboratory technicians around the world used their equipment in real settings.
Mapping global lab workflows to uncover usability gaps

A multinational tech company wanted to improve how game and app developers manage the business side of their products, particularly around monetization.
Streamlining developer tools for better business performance

A gaming company came to us because they needed to rewrite their privacy and safety messaging for kids.
Helping a gaming company talk to kids about online safety

A global technology company faced the of understanding how users form meaningful connections with AI chatbots and identifying the factors that lead to user attrition.
Shape global AI strategy through user-driven insights

An auto manufacturer needed to understand what car owners value most in their vehicles: what delights them, what frustrates them, and how they respond to new features.
Uncover what US drivers want from their in-vehicle experience

A healthcare company wanted to improve how technicians interacted with their hematology analyzers. Our findings gave the client a grounded view of what users needed.
Making lab tools easier to use for busy technicians

A global technology company needed to understand how users are onboarded into major tech ecosystems across international markets.
Compare global onboarding experiences across tech ecosystems

A pharmaceutical company was getting ready to launch a digital therapy app and needed to make sure it made sense in the local healthcare context and worked well for patients.
Localizing a depression therapy app for UK patients

A medical device manufacturer needed embedded HFE support to prepare regulatory documentation and conduct a validation study to demonstrate safe and effective use.
Supporting HFE documentation and validation for aesthetics device launch

The VA wanted to reduce the burden of managing chronic kidney disease (CKD) and ease the cognitive load on PCPs, who are often the first to detect the condition.
Supporting CKD care with better tools for PACT teams

The Human Systems Integration (HSI) division needed to clarify and promote the purpose of each of its programs in order to work more effectively across the organization.
Repositioning the VA’s human systems integration programs

A pharmaceutical company needed to understand how patients and caregivers administer oral medications using G-tubes and NG-tubes.
Identifying pain points in oral medication delivery for G- and NG-tube users

A digital payments provider wanted to understand how consumers would react to making purchases through smart speakers and uncover barriers to adoption.
Explore consumer adoption of smart speaker payment technology

A manufacturer of surgical and delivery devices wanted to redesign packaging and labeling for crash cart products to better align with clinical workflows.
Redesigning device packaging for safer clinical deployment
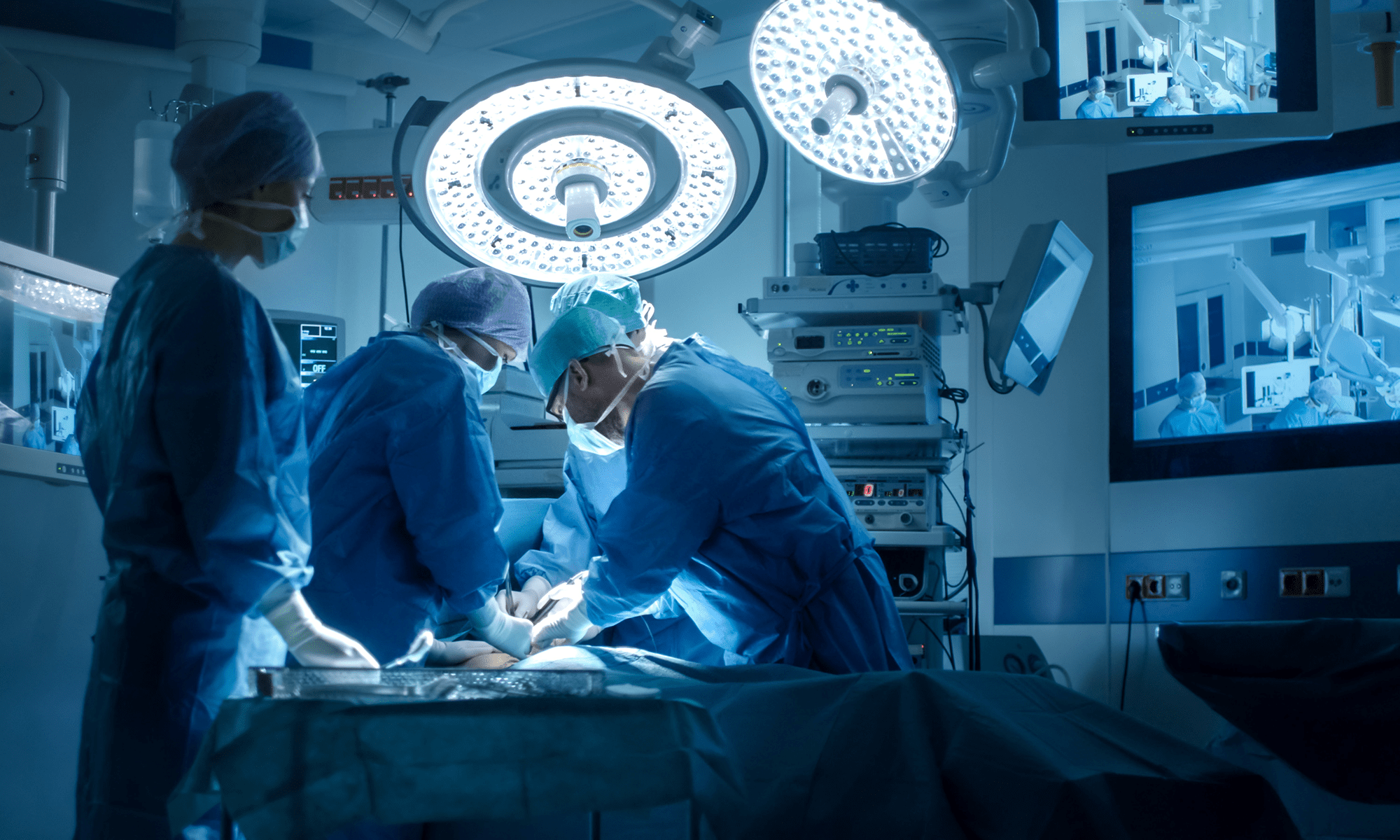
A surgical device manufacturer developed a next-generation mesh implant that offered clinical improvements but required changes to the implantation procedure.
Improving safety and clarity for surgical mesh procedures

A global consumer electronics company wanted to know how real users, especially older adults, experience onboarding and early use of their smartwatch.
Improve smartwatch onboarding and engagement across user groups

A global manufacturing company wanted to better understand the experiences of HVAC technicians during installation and service visits in customers' homes.
Improving the installation and service journey for HVAC technicians

Through multi-country cognitive interviews, we identified sources of user confusion and delivered culturally nuanced fixes that ensured clarity worldwide.
Identify sources of confusion across global markets for digital tool features

The United States Department of Veterans Affairs (VA) needed support optimizing the usability of its health information systems used across the Veterans Health Administration (VHA).
Improving VA health information systems

A women’s health company needed to assess its platform’s conformance to the European Accessibility Act (EAA) and Web Content Accessibility Guidelines (WCAG).
Improve accessibility compliance of menstrual tracking app

By revealing behavioral drivers of adoption, we helped a premium AI service refine onboarding, messaging, and features to secure confident market entry and growth.
Driving market success with human-centered AI

A pharmaceutical manufacturer needed to determine whether healthcare providers could safely and effectively administer a subcutaneous injection using an OBDS.
Evaluating safe and effective use of an on-body delivery system

Immersive research with call center reps and field technicians led to streamlined systems that reduced errors, cut costs, and improved customer satisfaction.
Transforming service operations through strategic UX partnership

We partnered with a pharmaceutical company to conduct in-lab testing with patients, mapping their ideal journey for a connected wearable drug delivery system.
Mapping the user journey for a connected wearable injection system
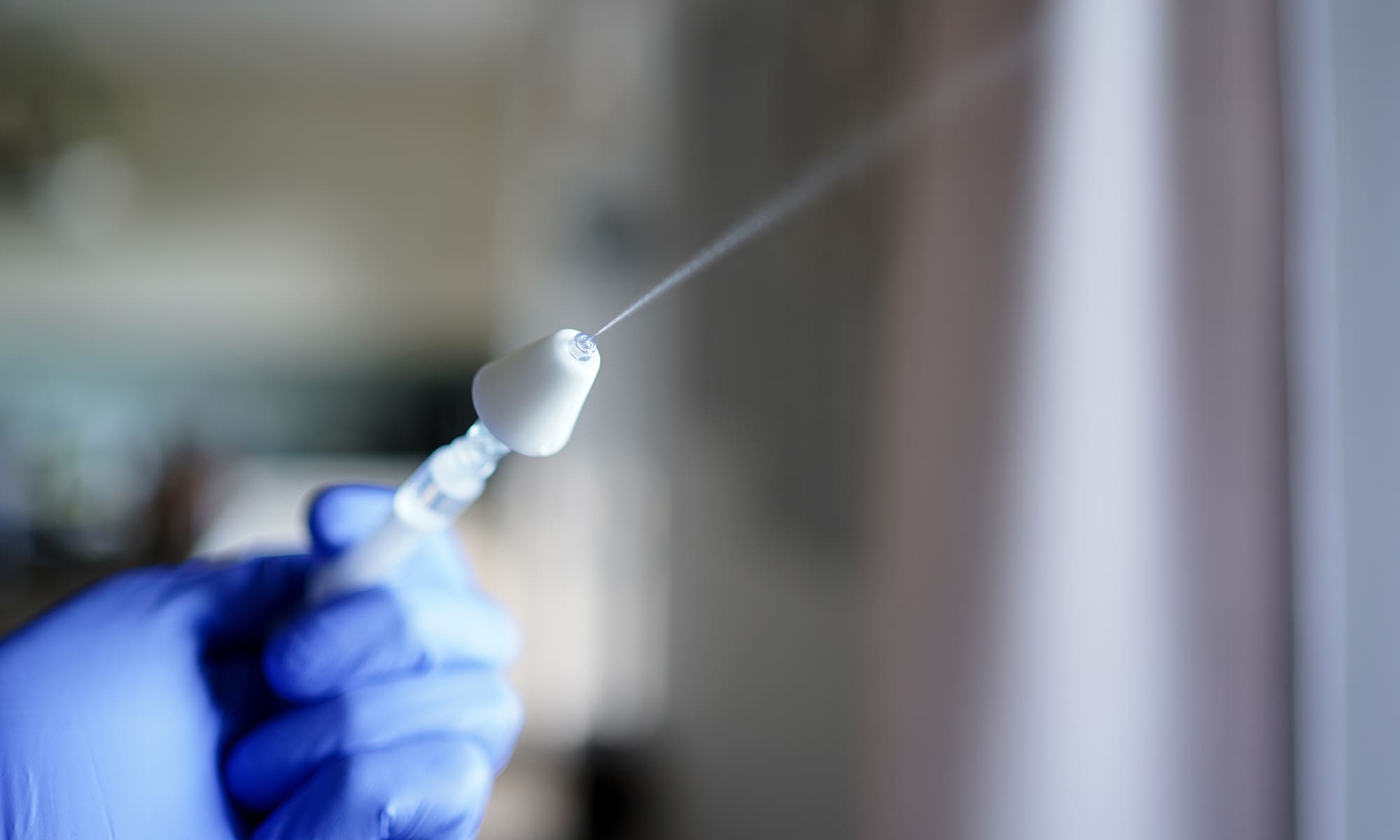
We helped a pharmaceutical manufacturer confirm that their emergency nasal MAD kit met safety and usability thresholds for regulatory approval.
Validate safe and effective use of emergency device

A pharmaceutical manufacturer was simultaneously improving an existing medication adherence system while developing a next-generation version with a companion mobile app.

















